Auf unserer Website kommen verschiedene Cookies zum Einsatz: Technisch notwendige Cookies verwenden wir zu dem Zweck, Funktionen wie das Login oder einen Warenkorb zu ermöglichen. Optionale Cookies verwenden wir zu Marketing- und Optimierungszwecken, insbesondere um für Sie relevante und interessante Anzeigen bei den Plattformen von Meta (Facebook, Instagram) zu schalten. Optionale Cookies können Sie ablehnen. Mehr Informationen zur Datenerhebung und -verarbeitung finden Sie in unserer Datenschutzerklärung.
-
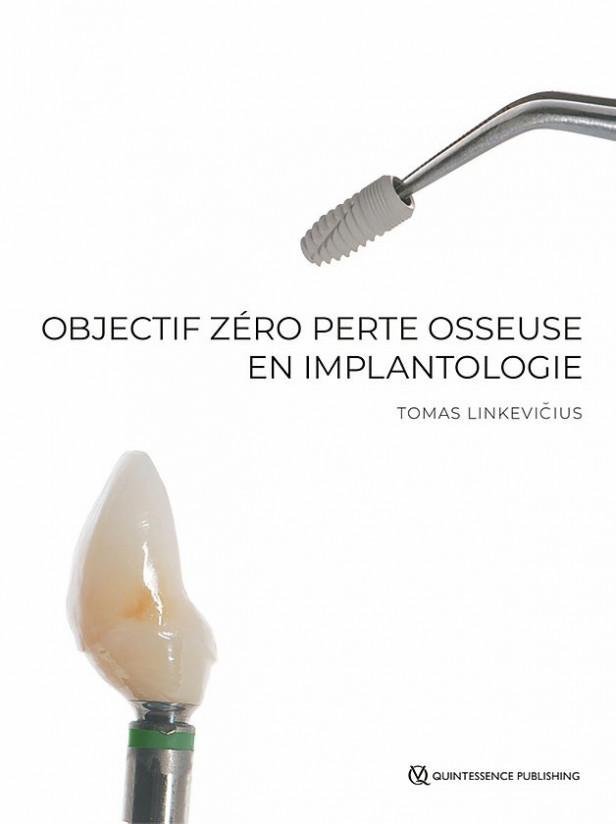
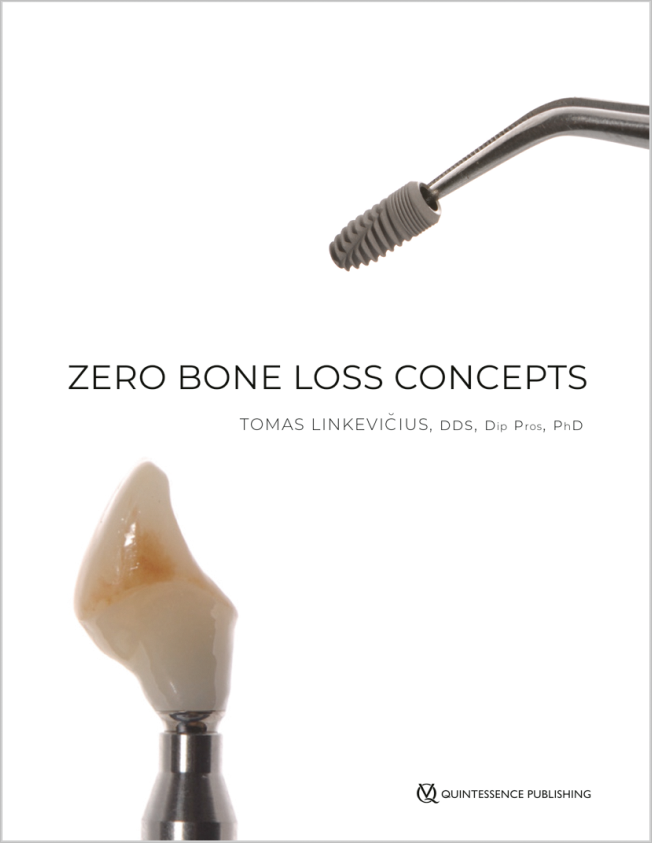
Bücher
-
Fachgebiete
- Akupunktur/Naturheilverfahren
- Allgemeinmedizin
- Anatomie
- Ästhetische Zahnheilkunde
- Dermatologie
- Endodontie
- Fachübergreifend
- Funktionsdiagnostik und -therapie
- HNO-Heilkunde
- Implantologie
- Kieferorthopädie
- Kinderbuch
- Kinderzahnheilkunde
- Literatur fürs Studium
- Medizingeschichte
- Mund-Kiefer-Gesichtschirurgie
- Oralchirurgie
- Parodontologie
- Patientenaufklärung
-
- Physiotherapie
- Plastische Chirurgie
- Praxismanagement
- Praxisteam
- Prophylaxe
- Prothetik
- Ratgeber Business & Karriere
- Ratgeber Elternschaft & Familienleben
- Ratgeber Fitness & Sport
- Ratgeber Gesundheit & Ernährung
- Ratgeber Gesundheit & Medizin
- Restaurative Zahnheilkunde
- Röntgenologie und Fotografie
- Seniorenzahnmedizin
- Sportmedizin
- Wissenschaft und Forschung
- Zahnerhaltung
- Zahnheilkunde allgemein
- Zahntechnik
-
Reihe
- Aesthetic Methods for Skin Rejuvenation
- Cell-to-Cell Communication
- Clinical Success
- Curriculum
- Die magischen Zahnfeen
- Dynamics of Orthodontics
- Facts - Das neue medizinische Nachschlagewerk
- ITI Treatment Guide Series
- Kompendium der dentalen Frästechnik
- QDT Yearbook
- QuintEssentials of Dental Practice
- Quintessenz Focus Zahnmedizin
- Zahnarzt | Manager | Unternehmer
-
Fachgebiete
-
Zeitschriften
- Allgemein
- Fachgebiete
-
Veranstaltungen
- Allgemein
- Fachgebiete
-
News
-
DVDs/eBooks/Apps
- Allgemein
- Fachgebiete
-
E-Learning
- Freie Zeitschriften
-
Reihen
- APW DVD Journal
- Cell-to-Cell Communication
- Compendium Implant-Supported Restorations
- Dental Technology on DVD
- Dental Video Journal
- Dental Video Magazin
- Dynamics of Orthodontics
- Kompendium Implantatprothetik
- Oral Surgery
- Periimplantäre Entzündungen
- Pioniere der Zahnmedizin: Funktion und Prothetik
- Pioniere der ZHK
- Quintessence Practice Live
- Quintessenz Praxis live
- Quintessenz Zahntechnik live
- Suturing Techniques
- Tabanella DVD-Compendium
- Zuhr-Hürzeler DVD-Kompendium
-
Partner
- Experten Hearings
-
Autoren
- Allgemein
-
Mehr
- Unternehmen
- Quintessenz
-
Verlage
-
Sprachen